Actors and definitions
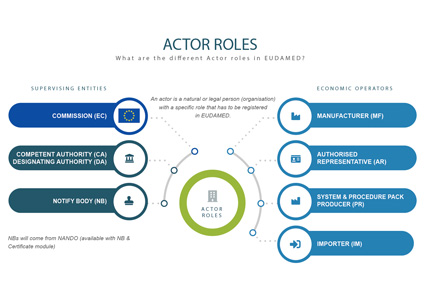
 INFOGRAPHIC: Actor ID/SRN and Actor roles
INFOGRAPHIC: Actor ID/SRN and Actor roles
Actors
Actors involved in EUDAMED are:
Supervising bodies
European Commission
Competent Authorities (CA), including Designating Authorities (DA)
Notified Bodies (NB)
Economic operators
Manufacturers - see Article 2(30) or Article 2(30) of Regulation 2017/745
System and procedure pack producers – any individual or organisation who places procedure packs and/or systems on the EU market
Authorised Representatives – see Article 2(32) of Regulation 2017/745
Importers – see Article 2(33) of Regulation 2017/745
Sponsors
see Article 2(49) of Regulation 2017/745
Definitions
Medical device – see Article 2(1) of Regulation 2017/745
In vitro diagnostic medical device – see Article 2(2) of Regulation 2017/746
Person responsible for regulatory compliance – see Article 15(1) and (3) of Regulation 2017/745
Single Registration Number (SRN) – see Article 31(2) of Regulation 2017/745