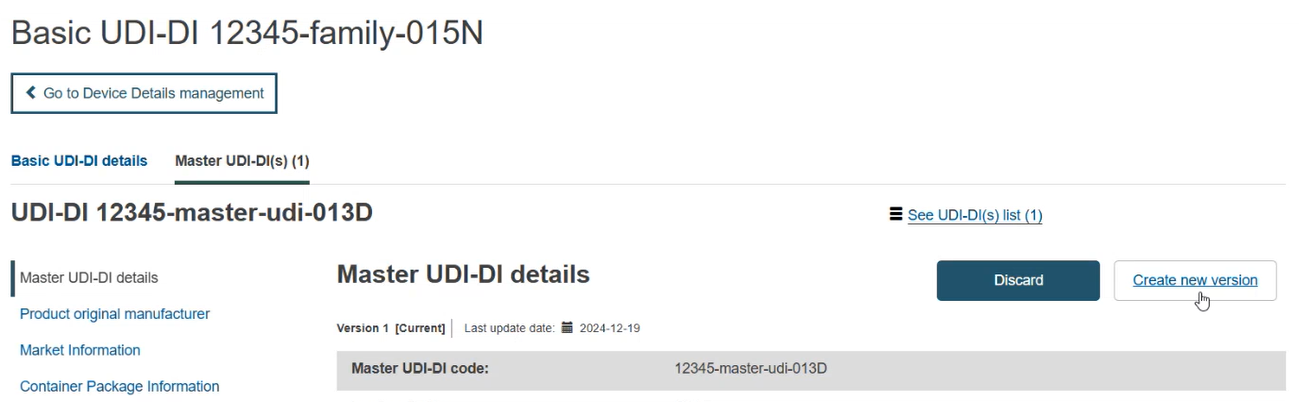
Update (create a new version) for UDI-DI/EUDAMED ID
 VIDEO: UDI assignment and updates
VIDEO: UDI assignment and updates
Follow the steps in section Manage your device UDI-DI/EUDAMED ID details to view a UDI-DI/EUDAMED ID.
Once inside the details of the selected UDI-DI, click on Create new version and proceed to update:

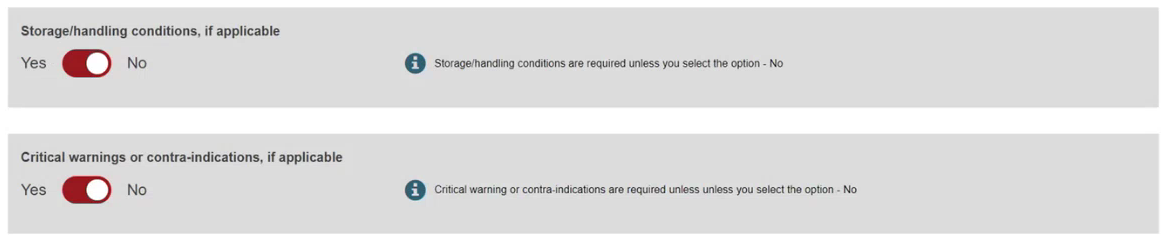
Note
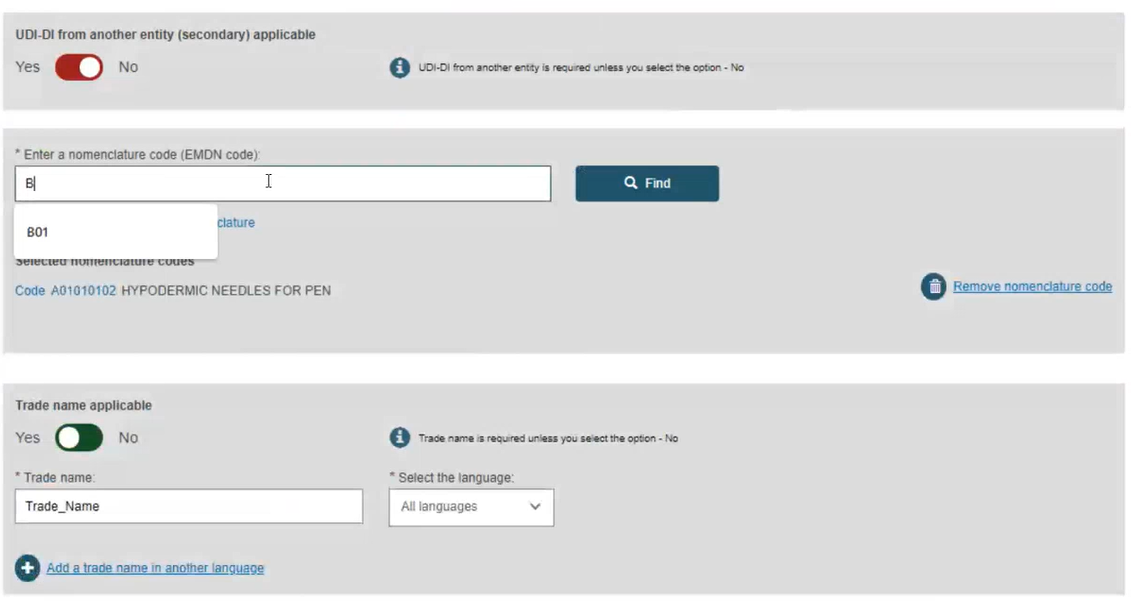
The available options for the UDI-DI status depend on the initial status of the device.
If the initial UDI-DI status of the device is either On the EU market or No longer placed on the EU market, when updating the UDI-DI status of the device, you can select either the On the EU market or the No longer placed on the EU market status.
If the initial UDI-DI status of the device is Not intended for the EU market, when updating the UDI-DI status of the device you can only select the On the EU market status.
Note
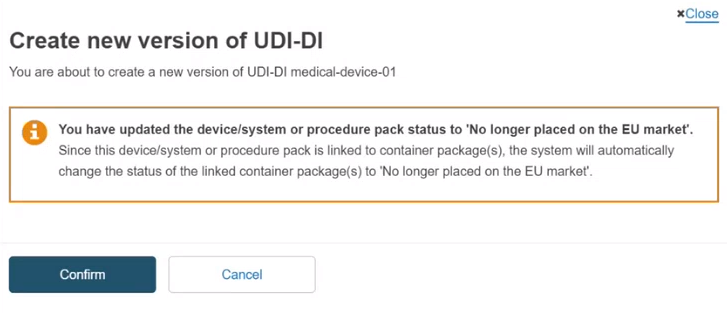
In the UDI-DI status field, if you select the No longer placed on the EU market status, the Market information will no longer be displayed and all container packages linked to this device will automatically be updated to the same status as the device.
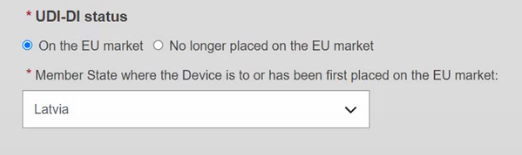
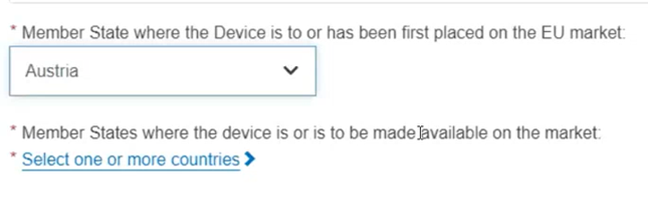
Otherwise, if you select the On the EU market status, you must select a Member State in the drop-down list where the device is or has been first placed on the EU market and the Member State(s) where the device is or is to be made available. You must also manually update all container packages linked to this device.
Tip
Master UDI-DI update variation
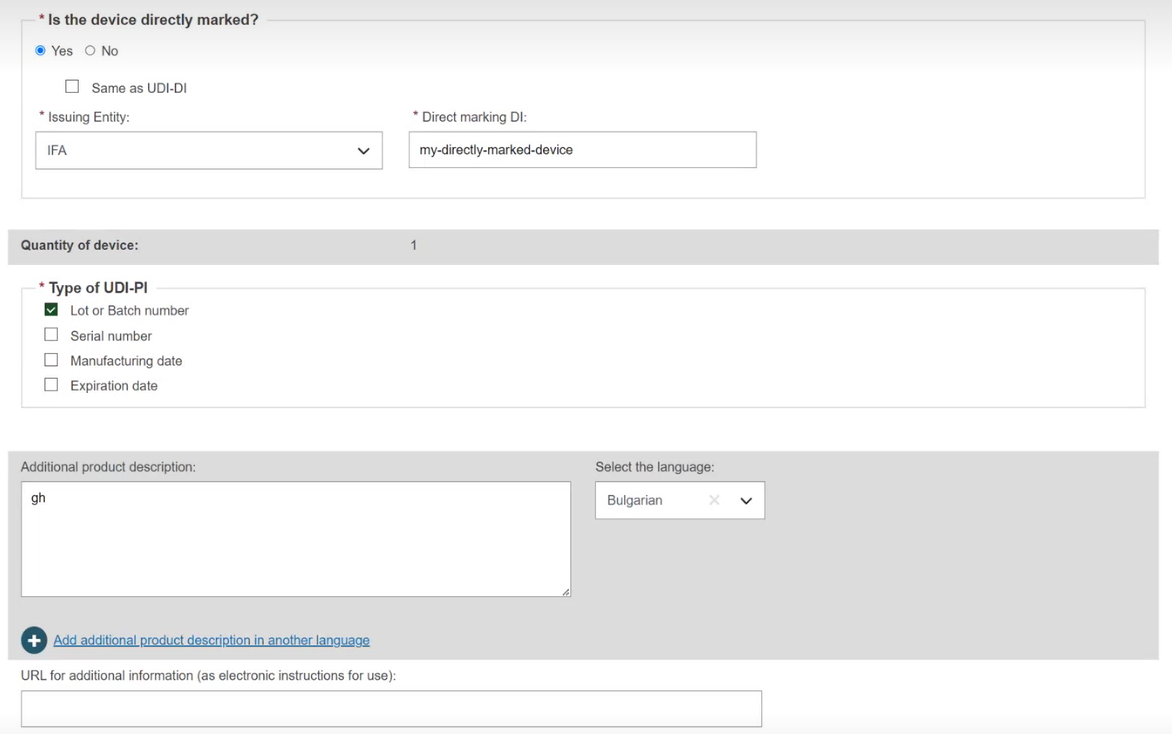
When creating a new version of a Master UDI-DI, the Quantity of device field is editable, whereas for the UDI-DI, it is not.

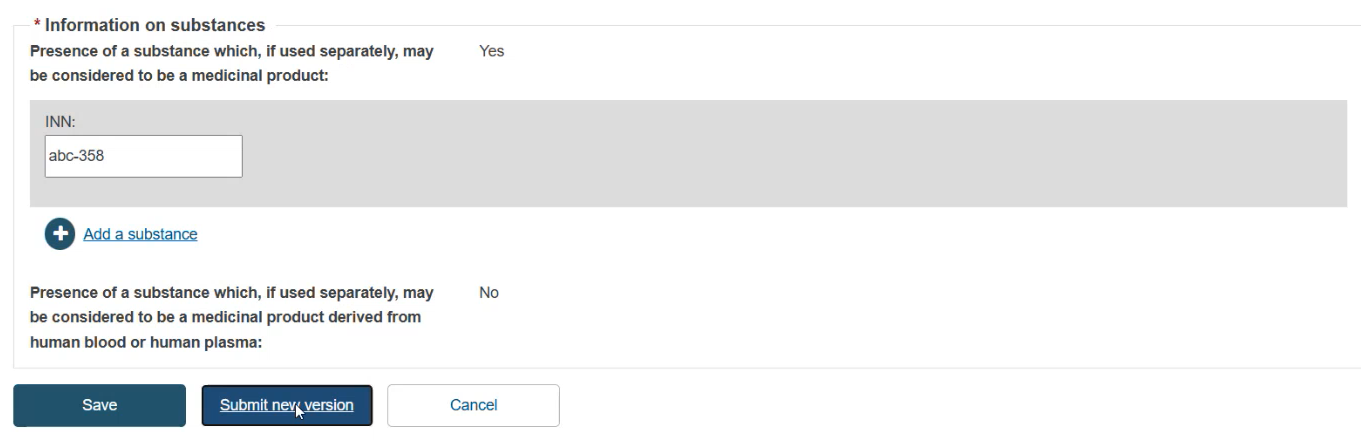
To finish the action you have two options:
Save to save the updated details without submitting the new version.
Submit new version, if you wish to finalise the update.
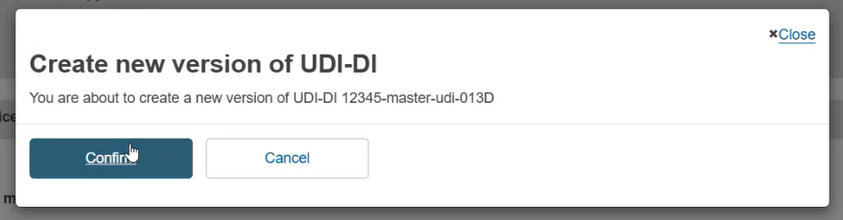
Once you have submitted the new version, click on Confirm in the pop-up window to finalise the update: