Register a not-followed CECP
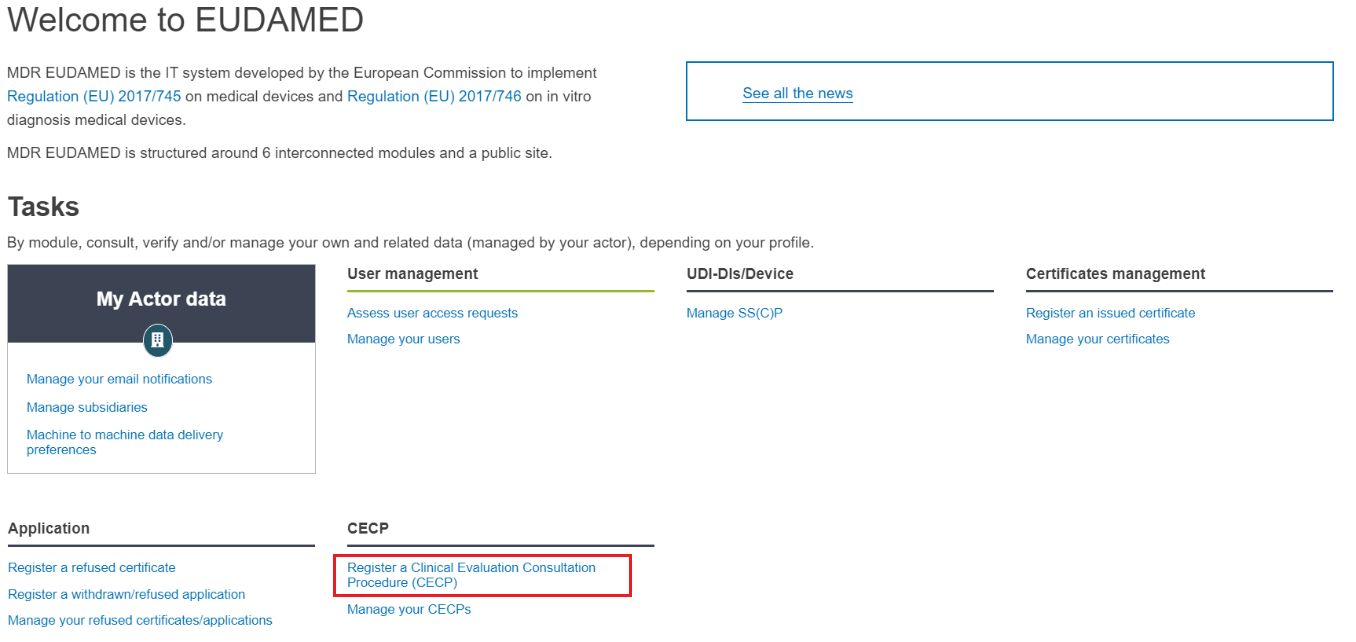
To register a not-followed CECP, look for the CECP section at the bottom and click on Register a Clinical Evaluation Consultation Procedure (CECP). You will be brought to the first step of the process:
In the first question Is this Clinical Evaluation consultation procedure followed? select No:
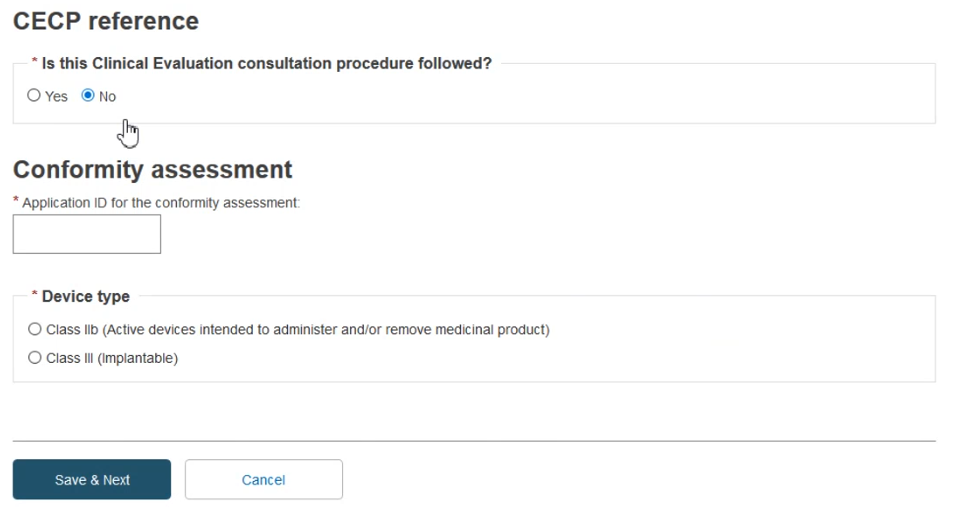
Enter the Application ID in the Conformity assessment and select a Device type from the two options available.
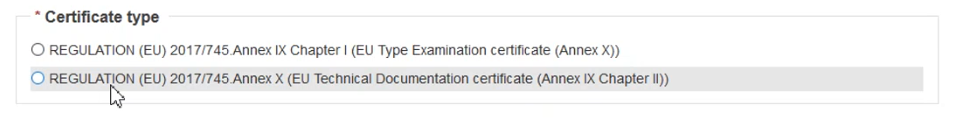
Once a device is chosen, a Certificate type field will pop-up. Then choose the correct regulation:
A new field will appear asking for the Actor ID/SRN number. Enter it, or part of it, and click Find:
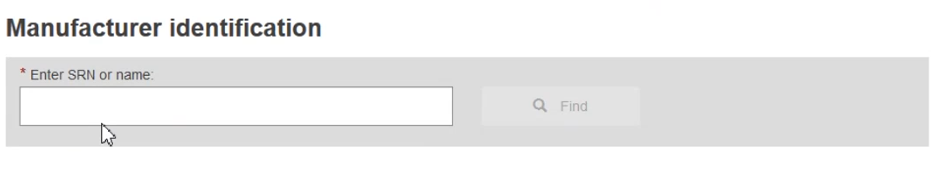
Once found in the list, select it and then click Save & Next:
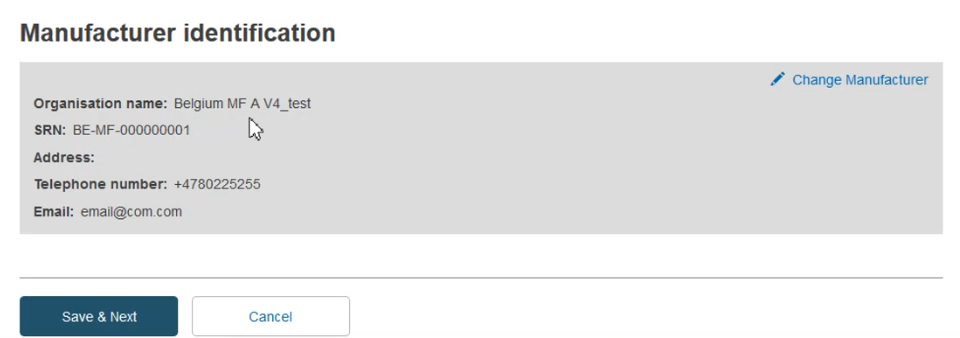
Follow all the steps in CECP record with Basic UDI-DI registered in EUDAMED page.
When you arrive to the Provision of CEAR document screen you will notice it is different from the usual window, with a new Reason for CECP not followed field:
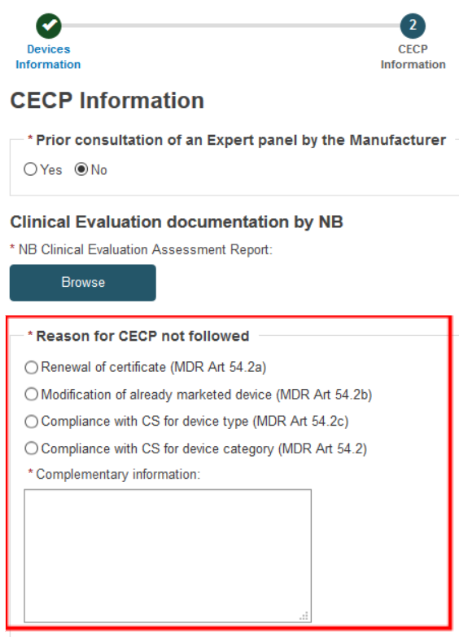
Select Yes or No and upload the NB Clinical Evaluation Assessment Report.
Select one of the reasons why the CECP is not followed and enter the complementary information.
Click Submit to finish, then click Submit my request. A confirmation screen will appear: