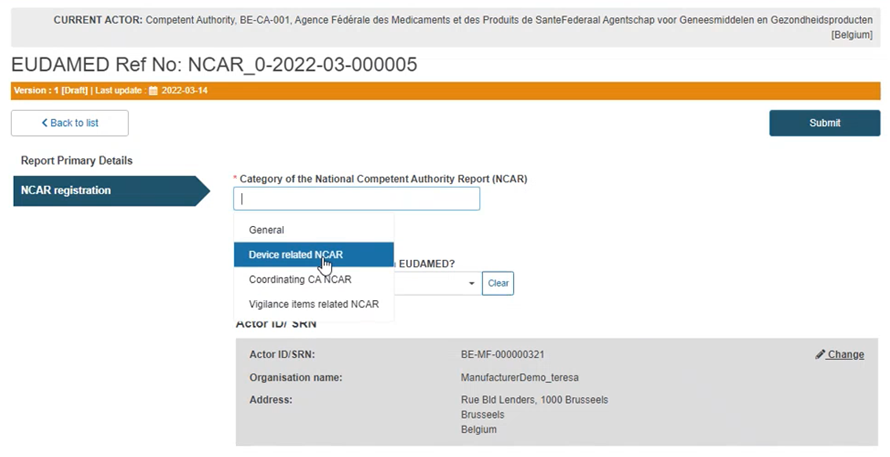
Case B: Device-related NCAR
Select the Device-related NCAR category in the first field:
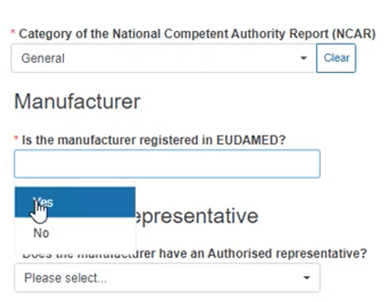
Select Yes or No in the Manufacturer field, as shown below:
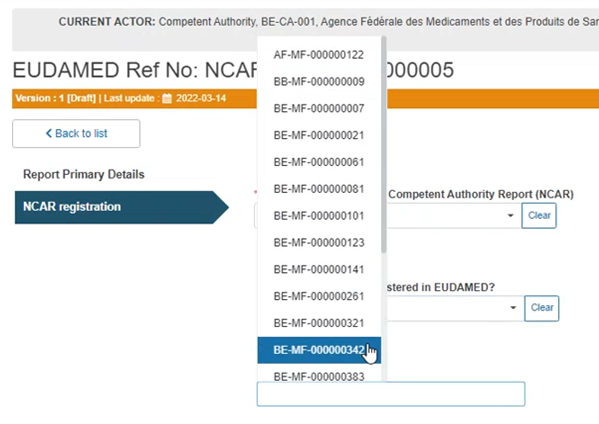
If you answered Yes to the previous question, you have to select the Actor ID/ SRN from the drop-down list in the field that will appear below:
Specify if the Manufacturer has an Authorised Representative:

Provide the Device information below:
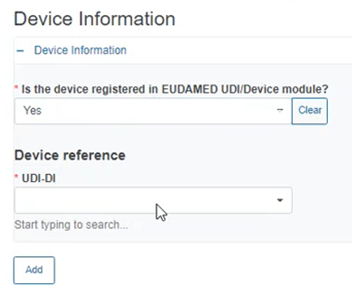
If you answer Yes, provide the UDI-DI of the device.
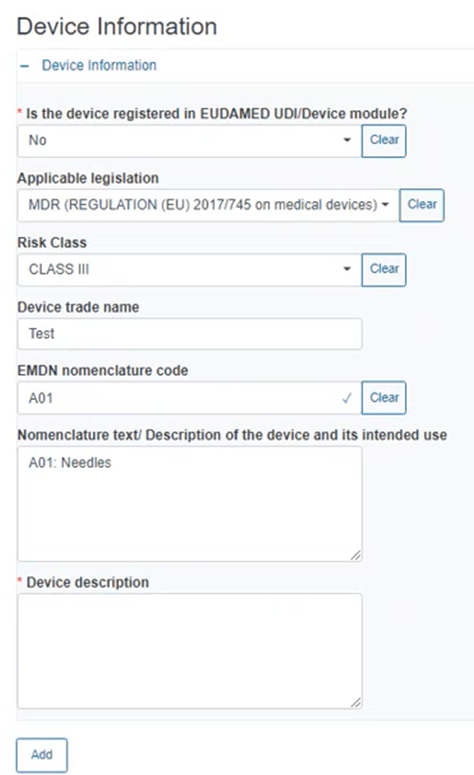
If you answered No to the above question, fill in the information in the appearing fields including Applicable legislation, Risk Class etc., as shown below:
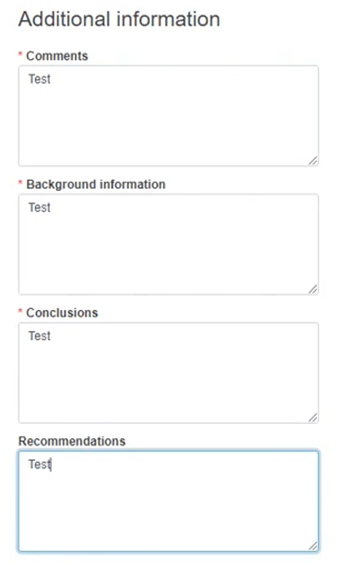
Provide any useful and relevant comments in the description fields under Additional information:
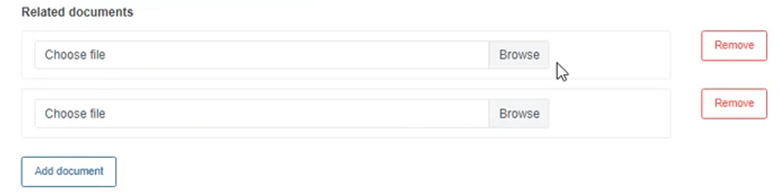
Click on Browse under Related documents to attach any documents relevant to the information provided:
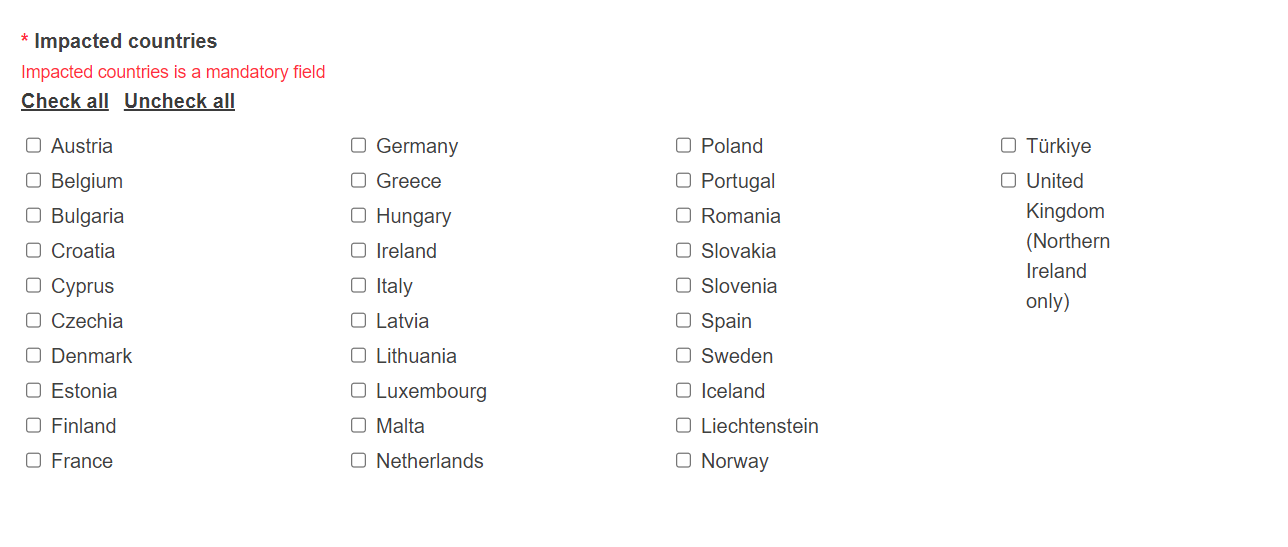
Complete this section by ticking the impacted country(/-ies), the Competent Authorities of which will be notified about this NCAR:
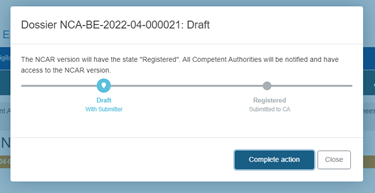
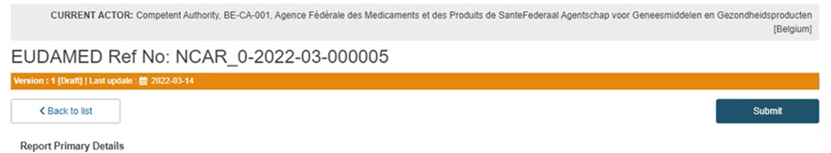
Submission:
Submit the report by clicking on the blue Submit button on the top right corner:
Finalise the submission of the report by clicking on Complete action in the pop-up window: