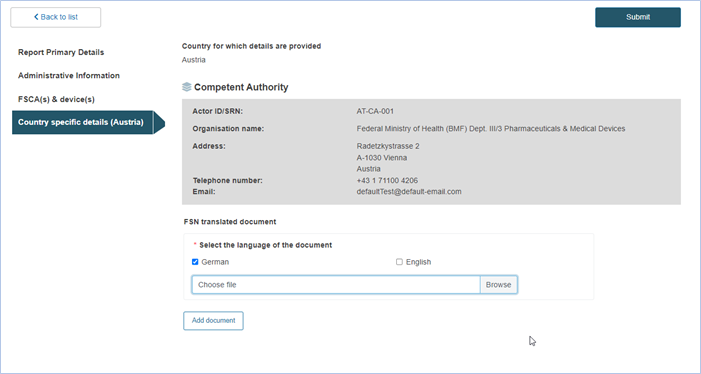
FSN submission
After having completed all previous steps, click on Submit at the top right corner of the screen to submit the FSN:
Tip
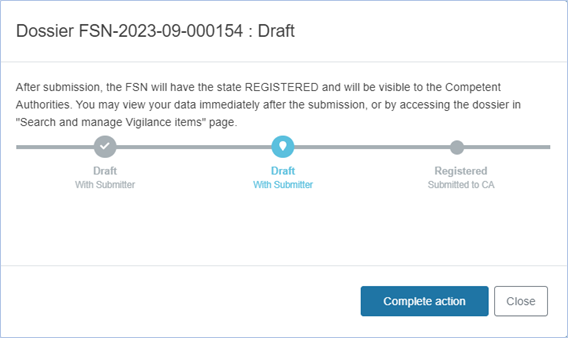
The system will confirm the submission via a pop-up dialogue box as follows:
Preliminary FSN: After submission, the preliminary FSN will be in registered state and visible to the Competent Authorities. CAs will be notified and will have two days to provide comments.
Final FSN: After submission, the final FSN will be in registered state and will also be publicly available on the public EUDAMED website. A sub-status for the referenced device(s) will be triggered (visible) in the EUDAMED Devices module.
Important
You may either proceed with the submission (Complete action button) or cancel the submission (Close).
Notification after submission
After submission, notifications will be issued as follows:
Preliminary FSN: action notification to the referenced CAs and information notification to all EOs/NBs.
Final FSN: information notification to the referenced CAs and to all EOs/NBs.